Age related macular degeneration (ARMD)
Pathophysiology
Age related macular degeneration (ARMD) is a current pathology of the portion of the retina responsible for the central and precise vision called the macula.
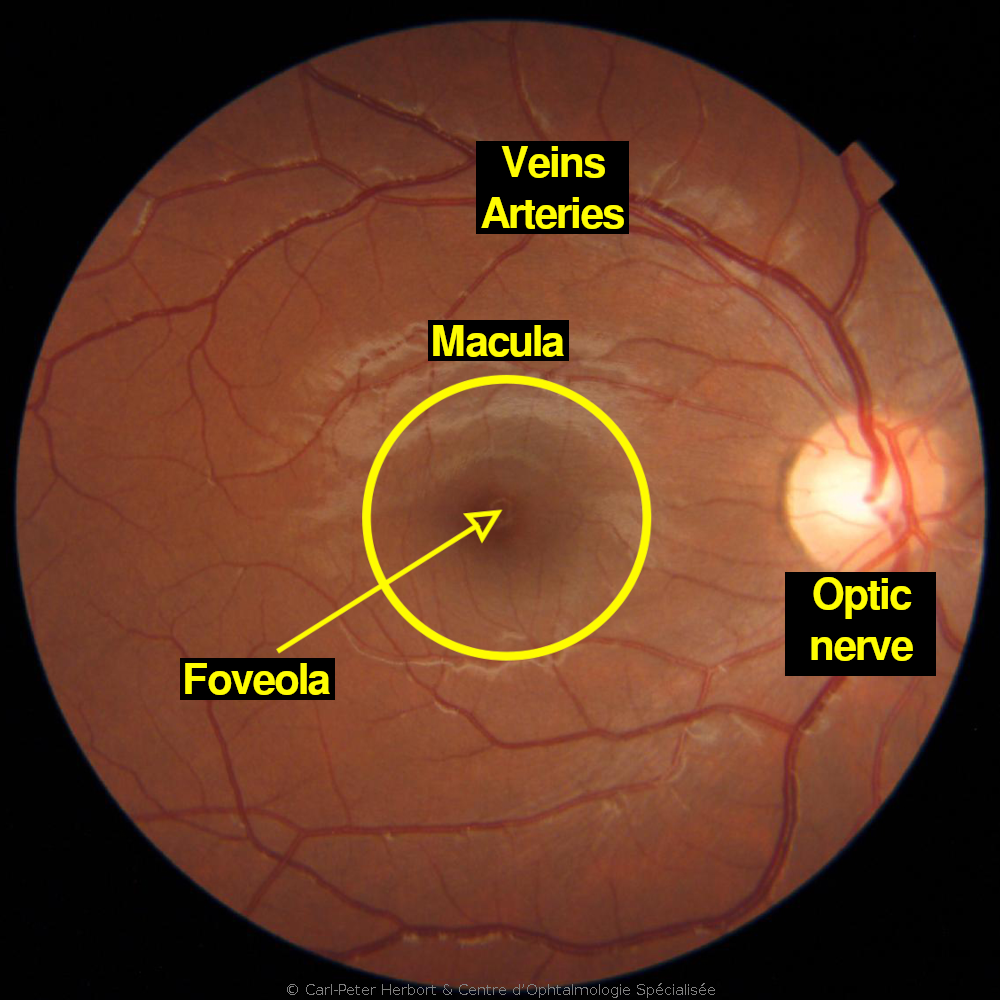
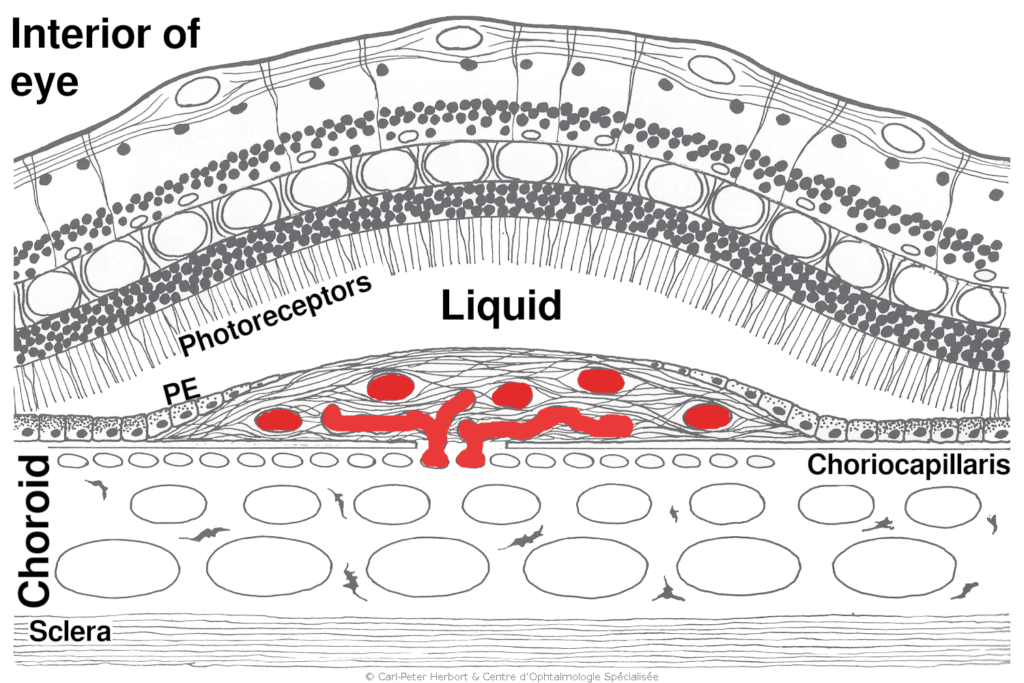
ARMD affects patients aged 60 and over and touches more often women. The origin is multifactorial. The development of the disease is caused by the accumulation of material in the cells of the pigmentary epithelium (PE) between the retina and the choroid. The cells of the PE have a function of “garbage removal”. Indeed, the constant activity of the photoreceptors during the vision process generates perpetually cellular debris which are eliminated by the cells of the PE. Underneath the retina, the choroid is a “lake” of blood providing oxygen and nutrients to the outer part of the retina. When the cells of the PE are no more capable to eliminate the cellular debris coming from the photoreceptors, they inflate with “garbage material” that accumulates within and outside the PE cells. The “garbage material” forms a barrier between the choroid and the retina to the passage of oxygen and nutrients to the retina. It forms Drusen (deposits), which lead to the degeneration of the outer part of the retina (dry ARMD). Sometimes the lack of oxygen makes the eye react and produce small “hormons” called Vascular Endothelial Growth Factor (VEGF) that promote the growth of new vessels (neovessels) which are fragile and at the origin of fluid leakage and distortion of the retina (wet or exudative ARMD). These neovessels can bleed.
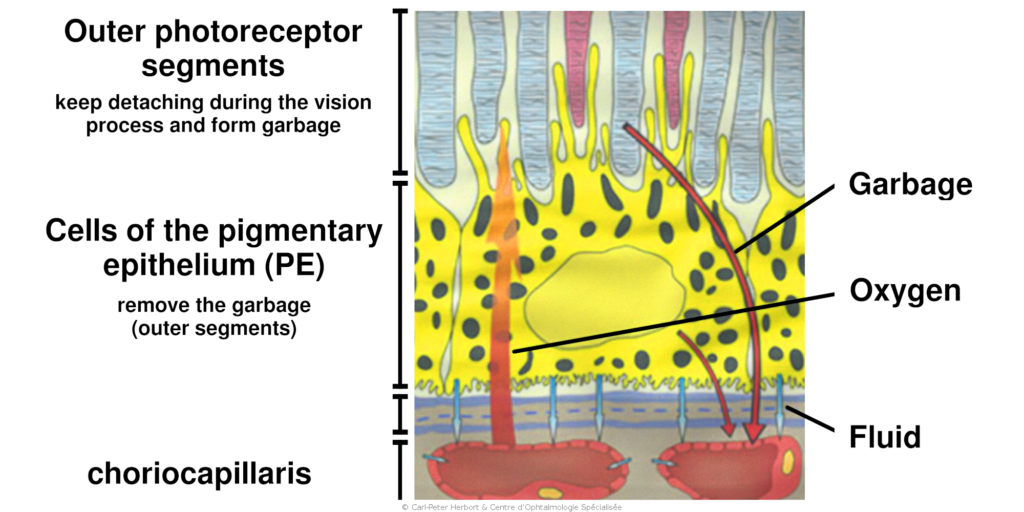
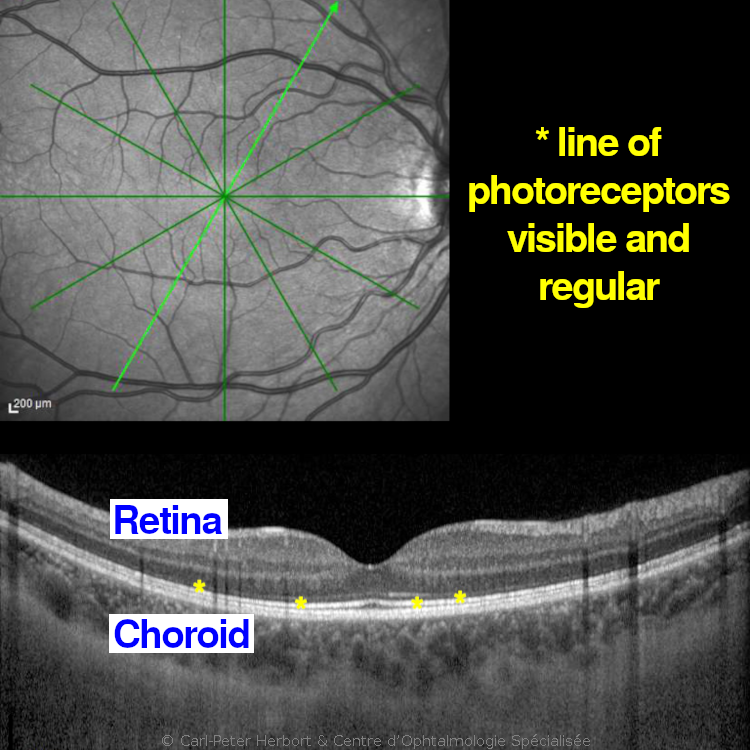
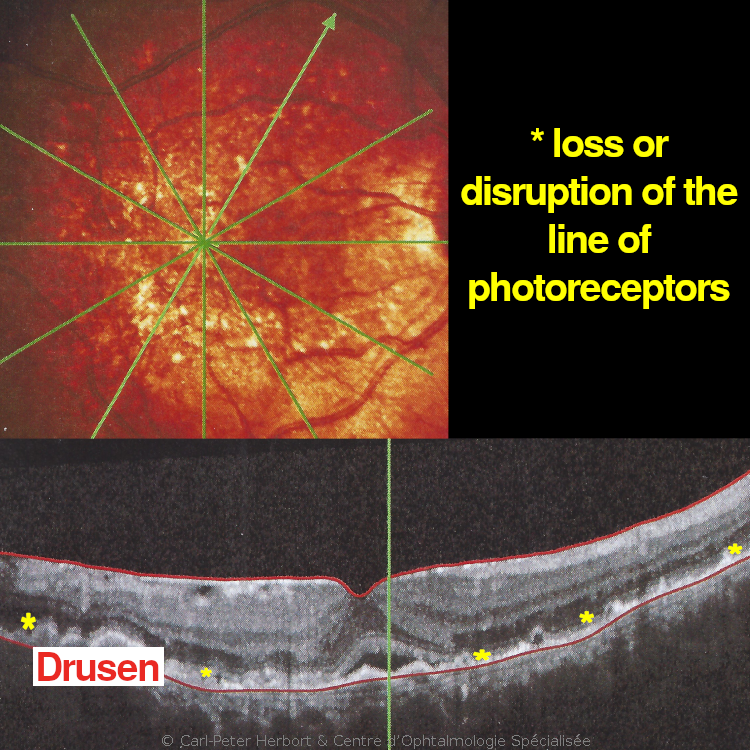
ARMD is henceforth divided into two categories, dry or atrophic ARMD and wet or exudative ARMD and their management is completely diverse.
Dry or atrophic ARMD
Atrophic ARMD is the more frequent form and no conclusive treatment is available. However, its evolution is slow. It is called atrophic because of areas of loss of retina or large geographic areas of loss of retina in advanced cases. To try to slow the progression, vitamin tablets can be given to support the retina but their effect is limited.
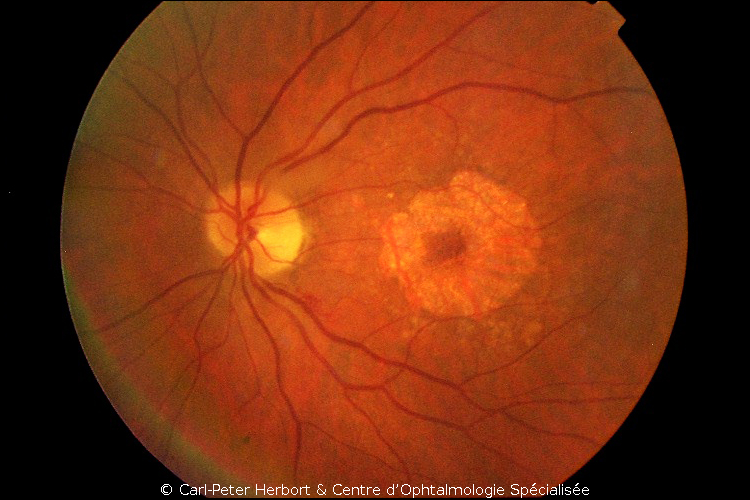
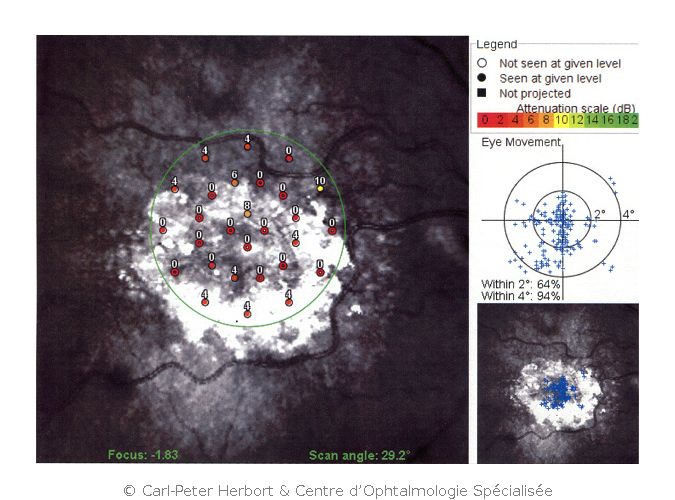
Wet or exudative ARMD
Wet or exudative ARMD, although not as frequent as dry ARMD, is more aggressive and can rapidly lead to loss of vision. It is called wet because leakage of fluid into the retina or subretinal space is the characteristic sign. The accumulation of fluid produces morphological alteration of the macula explaining the symptoms: the patients often describe a decrease of vision (mainly for reading), metamorphopsias (distortion of the image) or a dark area in the visual axis. The damage is caused by the development of abnormal neo-vessels that leak fluid across their imperfectly formed walls. Sometimes these fragile vessels can rupture and produce an intraretinal and/or subretinal bleeding. These neo-vessels develop because the retina signals its suffering and lack of oxygen by producing VEGF. Wet or exudative ARMD can be controlled by injecting intravitreally (inside the eye) an antibody that blocks VEGF and causes vessels to regress. This slows substantially the course of the disease.
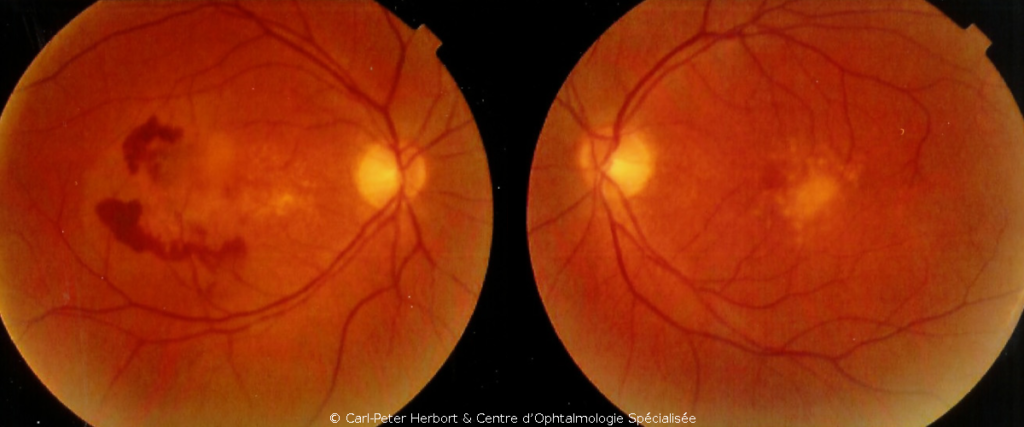
Historical note: intravitreal injections in Lausanne
The COS was one of two first ophthalmic centers which started intravitreal injections of anti-VEGF agents. The first treatment was made in March 2006. The substance was Avastin. Originally, Avastin was being used to treat cancer as it prevented the development of the vascularisation of the tumour. The first two patients were treated by using the intravenous administration of Avastin with the assistance of an oncologist. The following patients were treated by a direct injection into the eye of the substance. We had asked the Pharmacist Robert Golaz at “Pharmacie Internationale” to prepare aliquots for injection from the 100 mg vial which reduced the price of one treatment to CHF 100.- with an efficacy never obtained for the treatment of wet ARMD.
Of course, the pharmaceutical industry could not admit that such an efficient therapy was given away at such a ridiculous price. About 3 years later, a quasi-identical molecule was accepted by the drug regulatory agencies for ocular use and put on the market at the price of CHF 1800.- per treatment. This “new” substance had been tested for ocular use in studies for which the outcome was already known as the molecule was comparable to Avastin. As it had the ocular indication, doctors were legally obliged to use this twenty times more expensive treatment. Thereafter, we continued to use Avastin in a substantial proportion of cases, knowing that it was equally efficient when compared to the “new” substance. Very often, patients asked us to use the cheaper treatment. The inferiority of Avastin compared to the “new” substance was never established because there is none.
